Main navigation
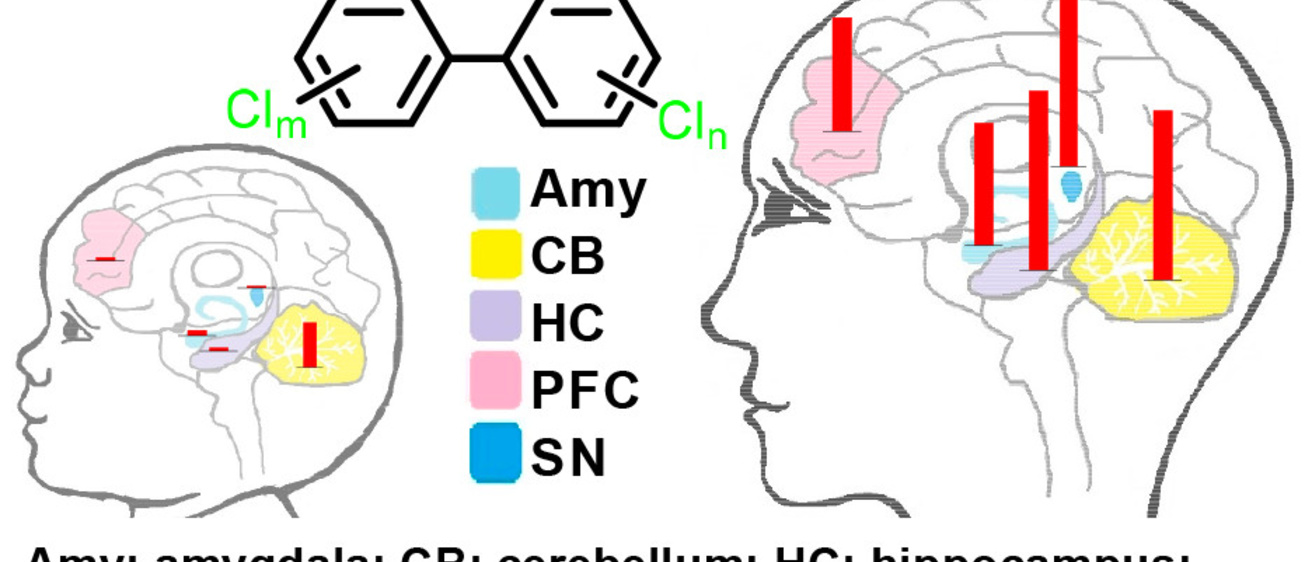
Project 1: Airborne PCBs and their metabolites: Risk factors for adverse neurodevelopmental outcomes in adolescence
Studies by the Iowa Superfund Research Program demonstrate that inhalation of indoor air, especially in U.S. schools contaminated with PCBs represents a current public health concern for U.S. adolescents. Although the adolescent brain is vulnerable to the toxicity of PCBs and their metabolites, information regarding the neurotoxicity of human metabolites of airborne PCBs—which differ significantly from those formed in rodents—is currently not available.
Goal
Assess and prevent neurotoxicity outcomes following exposure to PCB congener profiles detected in the air of U.S. schools and at PCB-contaminated Superfund sites.
Objective
Inform future risk assessment by defining the link between neurotoxic PCB metabolites present in the brain and neurotoxic outcomes following adolescent exposure.
Aims
- Determine the effects of an inhaled PCB mixture on biochemical markers and behavioral outcomes indicative of neurotoxicity in mice (collaboration with Project 2).
- Characterize the role of astrocytes and interactions with other cell types in PCB-mediated neurotoxicity in cells in culture.
2.1. Identify mechanistic targets of PCBs in primary mouse astrocyte cultures in vitro.
2.2. Assess the contribution of astrocyte interactions with other cell types (microglia and neurons) from mice on the adverse outcomes of PCBs in vitro
Project Team
Project leader

Hans-Joachim Lehmler, PhD
Co-investigators

Jonathan Doorn, PhD

Hanna Stevens, MD, PhD

Peter Thorne, PhD
Team members

Sean Brobston

Miguel Garcia Mares

Laura Gosse-Dean