Main navigation
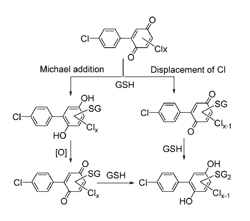
Polychlorinated biphenyls (PCBs) are hydroxylated by cytochrome P450 enzymes and can be further metabolized to PCB quinones. Further reactions of glutathione (GSH) with PCB quinones having different degrees of chlorination on the quinone ring were examined. Electron paramagnetic resonance (EPR) spectroscopy and LC-MS revealed two types of reactions yielding different products: (1) a non-enzymatic, nucleophilic displacement of chlorine on the quinone ring yielding a glutathionyl conjugated quinone; and (2) Michael addition of GSH to the quinone, a two-electron reduction, yielding a glutathionyl conjugated hydroquinone (Scheme 1).
The pKa of parent hydroquinone decreased by one unit as the degree of chlorination increased. This resulted in a corresponding increase in the oxidizability of these chlorinated hydroquinones. The reaction with oxygen appears to be first-order each in ionized hydroquinone and dioxygen, yielding hydrogen peroxide stoichiometrically. The generation of semiquinone radicals, superoxide, and hydroxyl radicals was observed by EPR; however, the mechanisms and yields vary depending on the degree of the chlorination of hydroquinone/quinone and the presence or absence of GSH. Our discovery that chlorinated quinones undergo a rapid, non-enzymatic dechlorination upon reaction with GSH opens a new view on mechanisms of metabolism and the toxicity of this class of compounds. (Nonenzymatic displacement of chlorine and formation of free radicals upon the reaction of glutathione with PCB quinones. Song Y, Wagner BA, Witmer JR, Lehmler HJ, Buettner GR. Proc Natl Acad Sci U S A. 2009 Jun 16;106(24):9725-30. Epub 2009 Jun 2. Open Access Article)