Main navigation
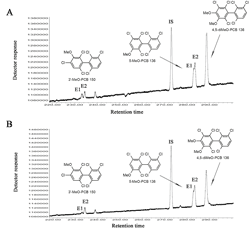
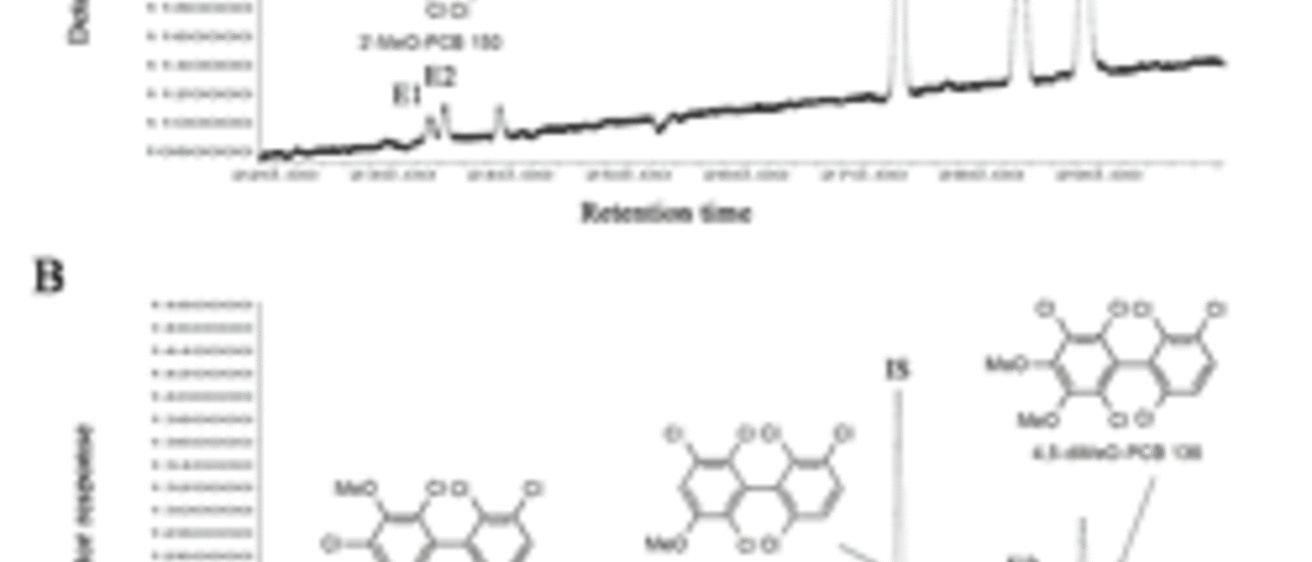
Several polychlorinated biphenyls (PCBs) and their hydroxylated metabolites display axial chirality. The Synthesis Core provided a series of chiral methoxylated PCB (MeO-PCB) standards to develop an enantioselective, gas chromatographic separation of MeO-PCBs using a chemically bonded -cyclodextrin column (Chirasil-Dex). The atropisomers of several MeO-PCBs could be separated on this column with resolutions ranging from 0.42-0.87 under isothermal or temperature programmed conditions. In addition, the enantiomeric fraction of OH-PCB 136 metabolites was determined in male and female rats treated with racemic PCB 136. As shown in the Figure, the second eluting enantiomer (E2) of 3’-MeO-PCB 150 and 5-MeO-PCB 136 was enriched in the liver of male (A) and female (B) Sprague-Dawley rats. Rats received two intraperitoneal injection of racemic PCB 136 (2 × 100 mol/kg body weight, 2 × 36.1 mg/kg body weight) dissolved in corn oil (5 ml/kg body weight) on day 1 and 4, and were euthanized on day 7 (IS = PCB 204 as internal standard) (I. Kania-Korwel et al.; J. Chromatogr. A 2008: 1207, 146-154 ).