Main navigation
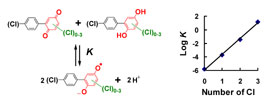
Polychlorinated biphenyls (PCBs) can be metabolized via hydroxylated and dihydroxylated metabolites to PCB quinone intermediates. We have recently demonstrated that both dihydroxy PCBs and PCB quinones can form semiquinone radicals (SQ·-) in vitro. These semiquinone radicals are reactive intermediates that have been implicated in the toxicity of lower chlorinated, airborne PCB congeners. To further investigate this observation, we synthesized a series of PCB hydroquinones and quinones with different degrees of chlorination of the (hydro-)quinone ring system. These PCB (hydro-)quinones readily react with oxygen or via comproportionation to yield the corresponding semiquinone free radicals, as detected by electron paramagnetic resonance spectroscopy. Surprisingly, the greater the number of chlorines on the (hydro-)quinone (oxygenated) ring, the higher the steady-state level of the resulting semiquinone radical at near neutral pH.
(Y. Song et al., J. Org. Chem. 2008:73, 8296-8304. DOI: 10.1021/jo801397g)